The activity of solutions can be estimated using concentration. Therefore, activity is used to tell chemists the difference between how many particles appear to be present in the solution, and the number of particles actually present in the solution. Now, when it comes to solids and liquids, we deal with concentrations. You found that the pressure for ethane as ideal gas was 24.47 atm, whereas the pressure for ethane as a real gas was 20.67 atm. Let's say that you were asked to calculate the pressure for one mole of ethane at 295.15 K behaving as an ideal gas (using the ideal gas law equation) and as a non-ideal gas (using the real gas equation). This can sound confusing, so let's look at an example. So, the activity of gas is considered to be the ratio of the actual partial pressure of the gas to its ideal partial pressure. When dealing with gases, they depend on pressure. If you want to learn more about gases and the ideal gas law, check out " Ideal Gas Laws"! Activities of Solids, Liquids, and GasesĪnother term that you need to be familiar with is the activity (α)of a species, chemists use the term activity to describe the deviation of ideal gases and solutions from ideal behavior. Molecules in these states of matter differ in their physical properties. Facts about Solids, Liquids, and GasesĮlements can exist in three states of matter: solids, liquids, and gases. 
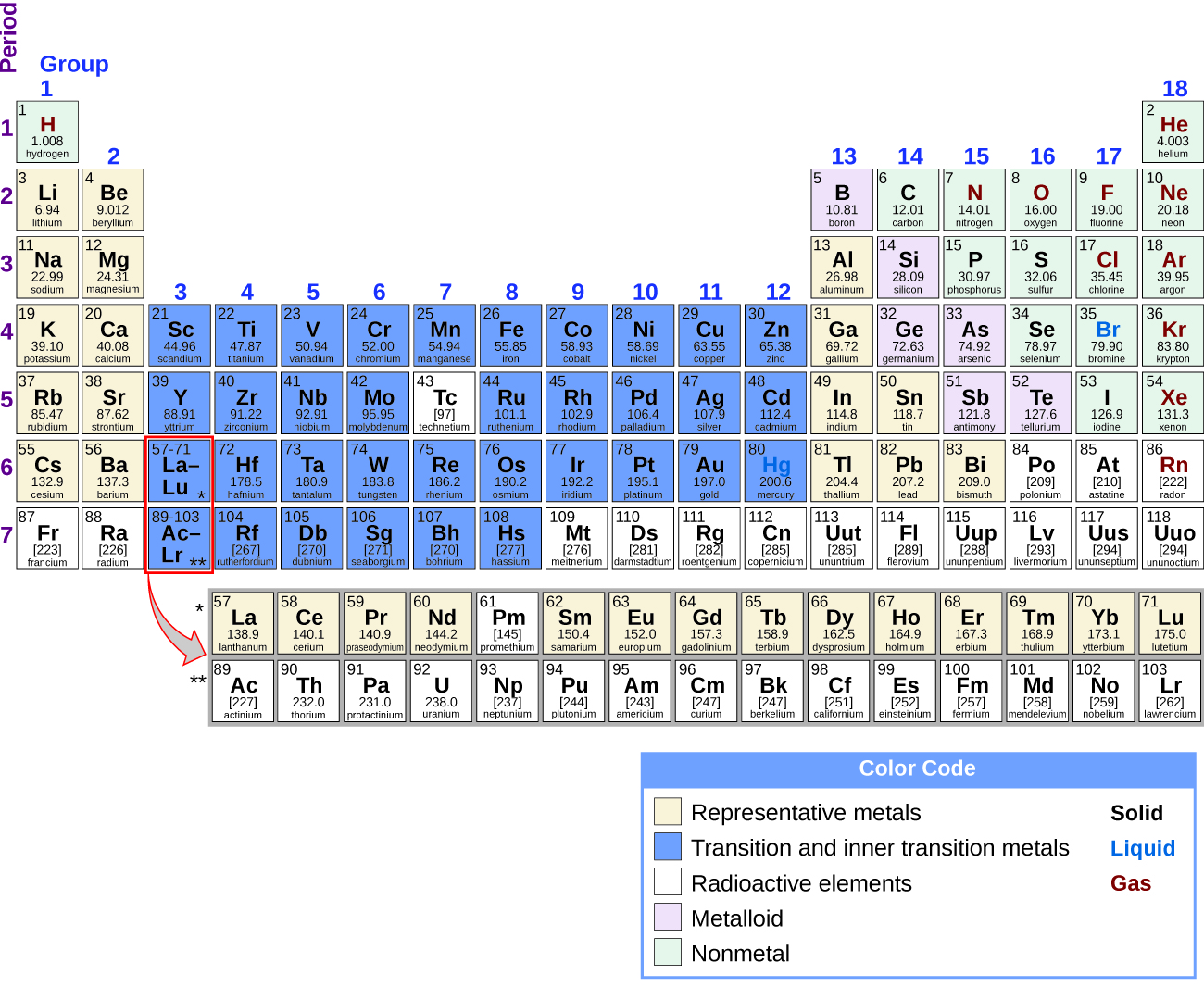
Now, let's dive into what solids, liquids, and gases are. Some nonmetals such as nitrogen, oxygen, fluorine, chlorine, and hydrogen are found as gases, whereas bromine and mercury are found in the liquid state. At room temperature (25 ☌) and under standard pressure (1 atm) conditions, most elements are found in nature in the solid state. Now, let's look at the periodic table, showing the state of the elements in nature. The simplest type of matter in chemistry is called an element, and an element is composed of only one type of atom! The functional unit of matter is an atom, at least for chemistry. Solids, Liquids, and Gases on the Periodic Tableīefore diving into the periodic table, you need to remember that matter is anything that has mass and occupies space. Lastly, we will compare solids, liquids, and gases.We will also talk about kinetic energy and chemical activity.Then, we will look at facts about solids, liquids, and gases.First, we will talk about where solids, liquids, and gases are found in the periodic table.Ice is a solid, water is a liquid and steam is a gas! But, what does this mean? Let's talk about liquids, solids and gases! These are all forms of the molecule H 2O, but in different phases, or states of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
